Pharma Stock Roundup: FDA Panel Endorses LLY's Donanemab, PFE's DMD Therapy Study Fails
This week, an FDA advisory committee unanimously recommended approval of Eli Lilly’s LLY Alzheimer's disease drug, donanemab. Pfizer’s PFE phase III study of investigational gene therapy, fordadistrogene movaparvovec, for ambulatory boys with Duchenne muscular dystrophy (DMD) failed to meet the primary endpoint as well as key secondary endpoints. The FDA approved AstraZeneca’s AZN type-II diabetes (T2D) medicine, Farxiga, for pediatric patients.AbbVie ABBV announced that it is in-licensing exclusive global rights to a next-generation TL1A antibody for the treatment of inflammatory bowel disease (IBD) from a Chinese biotech.
Recap of the Week’s Most Important Stories
FDA Panel Unanimously Endorses Lilly’s Donanemab: The FDA’s Peripheral and Central Nervous System Drugs Advisory Committee (PCNS) voted 11-0 unanimously recommending approval of Lilly’s Alzheimer's disease drug, donanemab. The committee said that data from clinical studies on donanemab demonstrated substantial evidence of efficacy and clinically meaningful slowing of disease progression for early symptomatic Alzheimer’s patients. Overall, the committee voted that the benefits of donanemab outweighed the risks despite some safety concerns. Anti-amyloid antibodies like donanemab can cause a brain swelling side effect called amyloid-related imaging abnormalities. However, to Lilly’s advantage, the panel said such risks could be addressed by “appropriate labelling and management”.
If approved by the FDA, donanemab will face strong competition from Biogen and Japanese partner Eisai’s Leqembi, which was granted full approval by the FDA for early Alzheimer’s disease in the United States in July 2023.
Pfizer’s Duchenne Therapy Study Fails: Pfizer’s phase III study, CIFFREO, evaluating its mini-dystrophin gene therapy fordadistrogene movaparvovec for treating Duchenne muscular dystrophy (DMD), failed to show improvement in motor function among patients four to eight years of age with DMD. The study failed to meet the primary endpoint of improvement in motor function, which was assessed by change in the North Star Ambulatory Assessment (“NSAA”) at one year after treatment. NSAA is a rating scale used to measure motor abilities. The study also failed to show a significant difference between participants treated with fordadistrogene movaparvovec and placebo for the key secondary endpoints. The study’s key secondary endpoints included 10-meter run/walk velocity and time to rise from floor velocity.
The CIFFREO study is currently on a dosing pause due to the death of a young boy who participated in another phase II study called DAYLIGHT in May this year. The DAYLIGHT study is evaluating fordadistrogene movaparvovec for treating DMD in boys aged two to three years.
FDA Approves AstraZeneca’s Farxiga for Pediatric Use: The FDA granted approval to AstraZeneca’s medicine, Farxiga, to improve glycaemic control in pediatric patients10 years and older with T2D. Farxiga is already approved for adults with T2D in several countries, including the United States and the EU. The approval for pediatric use was based on data from the T2NOW phase III study, which demonstrated a significant reduction in A1C, a marker of average blood sugar, in Farxiga-treated patients compared to placebo. Farxiga is a key top-line driver for AstraZeneca. The expanded approval for younger patients should further boost sales. Farxiga is already approved for pediatric patients aged 10 years and above with T2D in several countries, including the EU (marketed under the brand name Forxiga).
The FDA accepted AstraZeneca’s supplemental new drug application (sNDA), seeking expanded use of Tagrisso (osimertinib) for stage III EGFR-mutated non-small cell lung cancer (NSCLC) following treatment with chemoradiotherapy. With the FDA granting a priority review to the sNDA, a decision from the regulatory body is expected in the fourth quarter of 2024. If approved by the FDA, Tagrisso will be indicated for EGFRm NSCLC patients whose tumors have exon 19 deletions or exon 21 (L858R) mutations. The latest sNDA was based on data from the phase III LAURA study. Data from the study showed that treatment with Tagrisso reduced the risk of disease progression or death by 84% versus placebo.
AbbVie’s New Deal to Develop Novel IBD Treatments: AbbVie announced an exclusive global licensing deal with the Chinese company FutureGen Biopharmaceutical to develop FG-M701, a next-generation TL1A antibody for the treatment of IBD. IBD includes two conditions, Crohn's disease and ulcerative colitis both characterized by chronic inflammation in the gastrointestinal tract. FG-M701, a novel TL1A inhibitor, is in pre-clinical development. These next-generation IBD therapies are developed to drive greater efficacy and less frequent dosing than the first-generation TL1A antibodies. For the deal, AbbVie will pay $150 million in upfront and near-term milestone payments to FutureGen, with the latter also being entitled to an additional $1.56 billion in potential milestone payments.
The NYSE ARCA Pharmaceutical Index rose 0.5% in the last five trading sessions.
Large Cap Pharmaceuticals Industry 5YR % Return
Large Cap Pharmaceuticals Industry 5YR % Return
Here’s how the eight major stocks performed in the last five trading sessions.
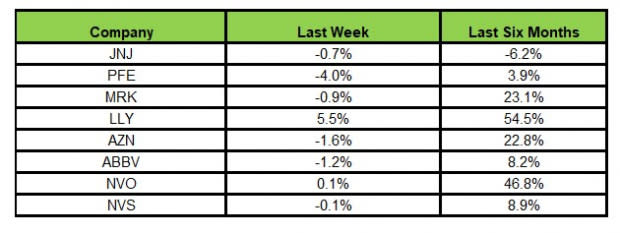
Image Source: Zacks Investment Research
In the last five trading sessions, Lilly rose the most (5.5%), while Pfizer declined the most (4.0%).
In the past six months, Lilly has risen the most (54.5%), while J&J has dropped the most (6.2%).
(See the last pharma stock roundup here:Pharma Stock Roundup: LLY’s CFO Departs, ABBV, AZN Drugs Get CHMP Approval)
What's Next in the Pharma World?
Watch this space for regular pipeline and regulatory updates next week.
Pfizer, Lilly and AstraZeneca have a Zacks Rank #3 (Hold) each. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
AstraZeneca PLC (AZN) : Free Stock Analysis Report
Pfizer Inc. (PFE) : Free Stock Analysis Report
Eli Lilly and Company (LLY) : Free Stock Analysis Report
AbbVie Inc. (ABBV) : Free Stock Analysis Report

 雅虎香港財經
雅虎香港財經 